FDA Approves Companion Diagnostic for Patients with Breast Cancer, Low HER2 Expression
The FDA approved a companion diagnostic to aid in the identification of patients with HER2-low metastatic breast cancer who may be candidates for treatment with fam-trastuzumab deruxtecan-nxki, according to a press release issued by Roche.
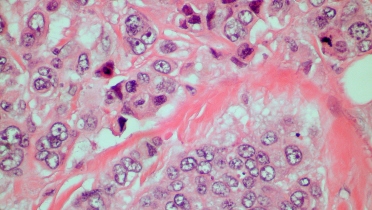
The PATHWAY anti-HER2 (4B5) Rabbit Monoclonal Primary Antibody Test uses a scoring algorithm designed to recognize patients with low expression of HER2, a receptor protein associated with cancer growth.
To date, it is the only FDA-approved companion diagnostic designed specifically to identify low HER2 expression.
Approval was based on data from the DESTINY-Breast04 study, which identified and treated patients based on low levels of HER2 expression.
The study reported a 50% reduction in the risk of death or disease recurrence, as well as a 6-month survival gain compared to standard of care for patients treated with fam-trastuzumab deruxtecan-nxki.
“Previously, metastatic breast cancer patients with a lower level of HER2 expression were considered to be part of the HER2-negative population and had no HER2-targeted treatment options. Now, they may be eligible for a HER2-targeted therapy, significantly increasing the number of patients who could have improved outcomes,” according to Thomas Schinecker, PhD, CEO of Roche Diagnostics, in the release
--
Reference:
https://www.roche.com/media/releases/med-cor-2022-10-04
Disclosures: Schinecker is an employee of Roche. DESTINY-Breast04 is being supported by Daiichi Sankyo and AstraZeneca.
Photo Credit: Getty Images.
By Cameron Kelsall, MD /alert Contributor









.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)


.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
_.jpg)


.jpg)
.jpg)
.jpg)
.jpg)

.jpg)

.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)

.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
 Featured Breast Cancer Videos
Featured Breast Cancer Videos.jpg)